胡迪醫(yī)學(xué) Hudi Medical Science

腫瘤精準(zhǔn)醫(yī)學(xué)20年的成就與挑戰(zhàn)
瀏覽量:4898 / 發(fā)布時(shí)間:2021-11-18
關(guān)注“胡迪醫(yī)學(xué)”,了解更多世界醫(yī)學(xué)動(dòng)態(tài)
胡迪主任參考世界頂級(jí)雜志《柳葉刀》
期刊社論,編譯了這篇推文,
讓大家了解腫瘤精準(zhǔn)醫(yī)學(xué)20年的發(fā)展,
展望精準(zhǔn)腫瘤學(xué)的發(fā)展前景。
腫瘤精準(zhǔn)醫(yī)學(xué)的成果和挑戰(zhàn)
20年前《人類(lèi)基因組計(jì)劃初稿》發(fā)表,
從此腫瘤精準(zhǔn)醫(yī)學(xué)駛進(jìn)了發(fā)展的快車(chē)道。
使用基因分型和基因組學(xué),
已經(jīng)成為一些癌癥標(biāo)準(zhǔn)治療的一部分,
這是腫瘤精準(zhǔn)診療的偉大進(jìn)步。
癌癥精準(zhǔn)醫(yī)學(xué)已經(jīng)給一部分病人
帶來(lái)了癌癥治愈,腫瘤分期減期,
明顯改善生命質(zhì)量,
延長(zhǎng)總體生存期等醫(yī)療結(jié)果,
這是生物學(xué)家和臨床醫(yī)學(xué)家們
共同努力,突破了部分癌癥難治的困境。
人們渴望、難以抗拒一種理想,
那就是能夠超越難以醫(yī)治的傳統(tǒng)界限,
實(shí)現(xiàn)更加精細(xì),更加有效,
“以病人為中心”的診療模式。
癌癥精準(zhǔn)醫(yī)學(xué)不是在路上,
而是已經(jīng)來(lái)到我們的身邊。
腫瘤精準(zhǔn)醫(yī)學(xué)的發(fā)展,
逐漸融合了大數(shù)據(jù)、蛋白組學(xué)、
轉(zhuǎn)錄組學(xué)、分子成像等領(lǐng)域技術(shù)。
盡管挑戰(zhàn),但是科學(xué)家和醫(yī)學(xué)家們
依然在將這種理想轉(zhuǎn)化為
有意義的、公平的醫(yī)療服務(wù)。
他們需要時(shí)間去解決一系列的、
因?yàn)榫珳?zhǔn)醫(yī)學(xué)而產(chǎn)生的新問(wèn)題,
比如如何消除醫(yī)療成本的增加,
如何推廣、設(shè)計(jì)充足的臨床試驗(yàn),
如何更有效地收集、整理各種數(shù)據(jù),
如何分析診療技術(shù)的醫(yī)療和經(jīng)濟(jì)價(jià)值,
如何規(guī)范和監(jiān)督精準(zhǔn)醫(yī)學(xué)的應(yīng)用、
如何解決帶來(lái)的公平和倫理相關(guān)的問(wèn)題。
什么是精準(zhǔn)醫(yī)學(xué)呢?
美國(guó)國(guó)家癌癥研究所(US NCI)
對(duì)精準(zhǔn)醫(yī)學(xué)的定義是,
一種利用個(gè)人基因、蛋白質(zhì)
和環(huán)境信息來(lái)預(yù)防、診斷和
治療疾病的醫(yī)學(xué)形式,但是,
利益相關(guān)者的定義大相徑庭。
靶向藥物治療臨床應(yīng)用于
許多實(shí)體癌和血液癌癥,
放療和手術(shù)的精確方法也是如此。
然而,即使有更成熟的基因檢測(cè),
轉(zhuǎn)化為臨床醫(yī)療遠(yuǎn)遠(yuǎn)落后于科學(xué)發(fā)現(xiàn)。
指南推薦的檢測(cè)經(jīng)常未得到充分使用,
并且因地區(qū)、種族和收入不同,
實(shí)際應(yīng)用也是有很大差異。
關(guān)注“胡迪醫(yī)學(xué)”,了解更多世界醫(yī)學(xué)動(dòng)態(tài)
精準(zhǔn)醫(yī)學(xué)面臨的挑戰(zhàn)有哪些?
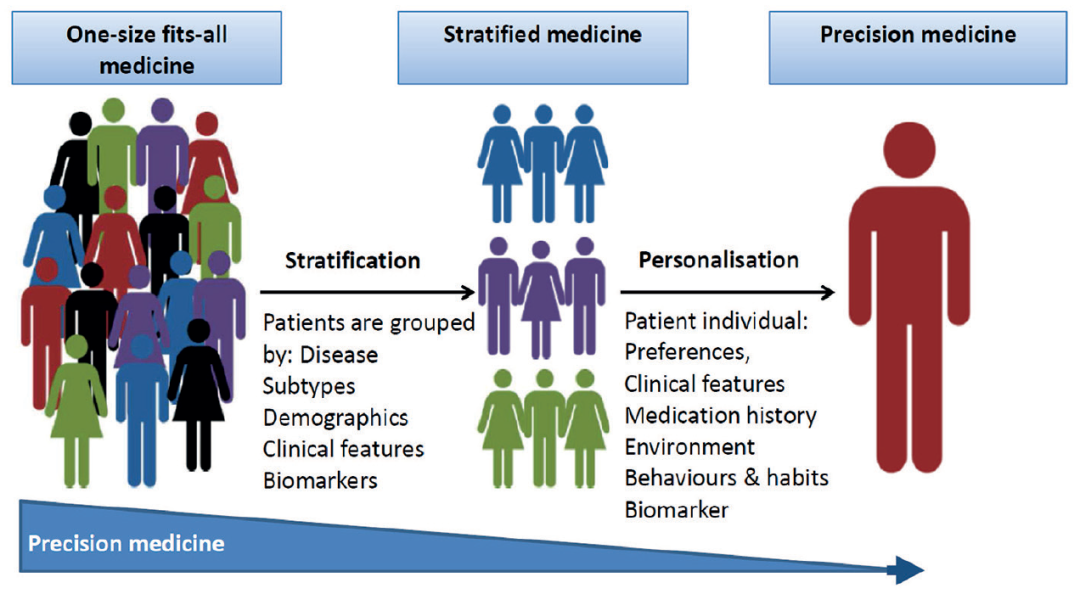
精準(zhǔn)醫(yī)學(xué)的許多進(jìn)展
尚未進(jìn)入臨床應(yīng)用的原因,
主要是臨床對(duì)精準(zhǔn)醫(yī)學(xué)的共同質(zhì)疑。
大家共同持有的想法認(rèn)為,
精準(zhǔn)醫(yī)學(xué)目前處于轉(zhuǎn)化階段,
缺乏標(biāo)準(zhǔn)的結(jié)果來(lái)定義臨床益處。
此外,
許多大型臨床試驗(yàn)缺乏足夠的比較對(duì)象,
而發(fā)展的快速度意味著許多試驗(yàn)
在報(bào)告時(shí)往往已經(jīng)過(guò)時(shí)了。
籃子、傘和其他自適應(yīng)設(shè)計(jì)的引入
是有幫助的,但增加了復(fù)雜性。
幫助指導(dǎo)臨床醫(yī)生選擇測(cè)試和
應(yīng)用臨床檢測(cè)結(jié)果時(shí),
缺乏很好的決策支持工具。
難以提供足夠的IT基礎(chǔ)設(shè)施,
也可能妨礙臨床使用。
競(jìng)爭(zhēng)激烈的市場(chǎng)和制藥公司的興奮點(diǎn),
使人們更關(guān)注技術(shù)突破而不是衛(wèi)生政策。
對(duì)精準(zhǔn)醫(yī)療實(shí)施的研究不足,
導(dǎo)致宣傳與患者實(shí)際療效之間存在差距。
如何解決精準(zhǔn)醫(yī)學(xué)的實(shí)施問(wèn)題?
針對(duì)癌癥精準(zhǔn)醫(yī)學(xué)帶來(lái)的光明前景,
目前已有幾個(gè)國(guó)家啟動(dòng)了國(guó)家舉措。
2015年,美國(guó)發(fā)起了“All of Us”項(xiàng)目,
該項(xiàng)目遠(yuǎn)遠(yuǎn)超出了癌癥診療的范疇。
該項(xiàng)目旨在招募100多萬(wàn)人,
從基因測(cè)序到醫(yī)療記錄,
收集使用10年以上數(shù)據(jù)。
澳大利亞基因組健康聯(lián)盟
是一個(gè)框架組織,
旨在改進(jìn)基因組學(xué)在臨床的應(yīng)用,
包括為臨床醫(yī)生提供
如何使用檢測(cè)結(jié)果的建議。
比利時(shí)、挪威、愛(ài)沙尼亞、法國(guó)和
以色列也制定了類(lèi)似方案。
關(guān)注“胡迪醫(yī)學(xué)”,了解更多世界醫(yī)學(xué)動(dòng)態(tài)
然而,這些項(xiàng)目中有許多試圖
協(xié)助實(shí)施基因組學(xué),
在提供現(xiàn)有技術(shù)教育的同時(shí),
可能難以整合最新的技術(shù)。
這些全國(guó)性的巨大努力
令人興奮,并吸引了人們的注意,
但風(fēng)險(xiǎn)是,在一些基礎(chǔ)研究實(shí)踐上,
科學(xué)家和臨床醫(yī)生之間缺乏外部合作和協(xié)議,
從而出現(xiàn)了多重努力,
阻礙了對(duì)患者的好處。
與許多新的醫(yī)療方法一樣,
高收入國(guó)家和低收入國(guó)家之間
在獲得新醫(yī)療方面存在不平等。
這種差距基于一種錯(cuò)誤的信念,
即低收入和中等收入國(guó)家需要
花費(fèi)太多資源,來(lái)實(shí)施這些新型的技術(shù)。
精準(zhǔn)醫(yī)療代表著一系列層次的技術(shù),
包括大數(shù)據(jù)和基因組測(cè)序。
巴西、中國(guó)和印度等國(guó)家
在這方面已經(jīng)擁有很多經(jīng)驗(yàn)和能力。
利用基因檢測(cè)和大數(shù)據(jù)能夠
更有效地利用現(xiàn)有資源,
可以在低收入或中等收入地區(qū)
通過(guò)合理的投資和優(yōu)先次序
獲得切實(shí)的成果,
但到目前為止,
這種情況還沒(méi)有發(fā)生。
一種更精確的藥物,
在減少不良事件的同時(shí)提高治療效果,
當(dāng)然是我們希望達(dá)到的目標(biāo)。
過(guò)去20年大家只是受到
先進(jìn)科學(xué)概念上突破的影響,
但是,沒(méi)有對(duì)實(shí)施精準(zhǔn)醫(yī)學(xué)
需要的基礎(chǔ)設(shè)施
和對(duì)病人醫(yī)療帶來(lái)的實(shí)效性,
給予足夠的關(guān)注。
精準(zhǔn)腫瘤學(xué)將繼續(xù)提供科學(xué)進(jìn)步,
但要為患者創(chuàng)造有意義的診療變化,
需要國(guó)際間的多種多樣的合作研究,
并為病人提供超越實(shí)驗(yàn)室的整體治療方法。
癌癥診療體系建設(shè)遠(yuǎn)遠(yuǎn)超越了
癌癥治療的本身。
關(guān)注“胡迪醫(yī)學(xué)”,了解更多世界醫(yī)學(xué)動(dòng)態(tài)
聲明:本文系健瑞寶醫(yī)療整理創(chuàng)作,如需轉(zhuǎn)載請(qǐng)取得許可,轉(zhuǎn)載時(shí)注明出處。本文屬醫(yī)療技術(shù)講座性質(zhì),不應(yīng)被視為任何醫(yī)療建議或治療方案,如您需要,請(qǐng)咨詢(xún)執(zhí)業(yè)醫(yī)師。刊載上述內(nèi)容,“胡迪醫(yī)學(xué)”尊重信息出處的原意,對(duì)文中陳述、觀(guān)點(diǎn)和判斷保持中立,不對(duì)所包含內(nèi)容的準(zhǔn)確性、可靠性或完整性提供任何明示或暗示的保證。如果您認(rèn)為文章內(nèi)容或者來(lái)源標(biāo)注與事實(shí)不符,請(qǐng)告訴我們,我們將與您積極協(xié)商解決。謝謝大家關(guān)注"胡迪醫(yī)學(xué)"。
文章中圖片和相關(guān)數(shù)據(jù)來(lái)自互聯(lián)網(wǎng),圖片及資料版權(quán)歸原作者,如有異議,請(qǐng)及時(shí)和我們聯(lián)系,我們盡快協(xié)商解決。

引用柳葉刀期刊社論的來(lái)源:
EDITORIAL| VOLUME 397, ISSUE 10287, P1781, MAY 15, 2021
20 years of precision medicine in oncology
The Lancet
DOI:https://doi.org/10.1016/S0140-6736(21)01099-0
In the 20 years since the publication of the first draft of the human genome project, the use of genotyping and genomics have become part of standard treatment for some cancers. The desire to go beyond blanket, and often difficult, treatments for patients to a more refined, efficient, and patient-centred approach is an ideal that is hard to oppose. But as precision medicine in oncology expands to include big data, proteomics, transcriptomics, molecular imaging, and more, there are serious challenges ahead to translate that ideal into meaningful and equitable health care for patients. Issues surrounding rising costs, adequate clinical trial design and data, regulation, defining meaningful benefits to patients, and equity remain to be solved.
The US National Cancer Institute's definition of precision medicine is “a form of medicine that uses information about a person's genes, proteins and environment to prevent, diagnose and treat disease”, although stakeholders' definitions vary widely. Targeted drug therapy is in clinical use for many solid and haematological cancers, as are precision approaches to radiotherapy and surgery. However, even for more established genetic tests, translation into clinical care has lagged behind scientific discovery. Guideline-recommended testing is often underused and varies by region, race, and income.
Many advances in precision medicine have not made it into the clinic because of common challenges to the field. For ideas currently in the translational phase, there is a lack of standard outcomes to define clinical benefit. In addition, many large clinical trials lack adequate comparators and the pace of development means many are often out of date by the time they report. The introduction of basket, umbrella, and other adaptive designs are helpful but add complexity. A lack of decision support tools to help guide clinicians with the choice of tests and application of results, and difficulty with providing sufficient IT infrastructure, may also hamper clinical use. A highly competitive market and excitement from pharmaceutical companies puts a focus on technological breakthrough rather than health policy. Insufficient research into the implementation of precision medicine has resulted in a gap between the hype and the material gains in outcomes for patients.
Several national initiatives to deliver on the promise of precision medicine in oncology exist. In 2015, the USA initiated the All of Us project, which goes far beyond cancer care. This project is designed to enrol more than a million people and use data over 10 years, from genetic sequencing to health-care records. The Australian Genomics Health Alliance is a framework that aims to improve the translation of genomics into the clinic, including advice for clinicians on how to use results. Programmes are also in place in Belgium, Norway, Estonia, France, and Israel. However, many of these programmes try to assist with implementing genomics and might have difficulty incorporating newer technologies while providing education on established techniques. These vast national efforts are exciting and attract attention, but there is a risk that insufficient external collaboration and agreement among scientists and clinicians on some of the basic research practices replicates efforts and hinders the benefits to patients.
As with many new medical treatments, there is inequity in access between high-income and low-income countries. The determinism that is relied on to justify this disparity, based on a belief that some newer forms of technology require too many resources for low-income and middle-income countries (LMICs) to implement, is a fallacy. Precision medicine represents a hierarchy of technologies, including big data and genomic sequencing, which many LMICs—eg, Brazil, China, and India—already have experience in and capacity for. Use of genetic testing and big data to more efficiently use existing resources could see tangible results in these regions with reasonable investment and prioritisation. But thus far, this has not occurred.
A more precise medicine, which minimises adverse events while enhancing therapeutic impact, is of course the desired goal. But the past 20 years have been coloured by advanced scientific conceptual breakthroughs without adequate focus on the basic building blocks of implementation and the practicalities of patient care. Precision oncology will continue to deliver scientific advances, but to create meaningful changes for patients, international collaborative research and a holistic approach to the patient that goes beyond the lab are required. Cancer care is much more than cancer treatment.
關(guān)閉
